We recently received this video clip from one of our Consultant Training Program students, Bhavya Sharma. It shows a microbe undergoing a process you’d think we might witness more often, working as we do with things like compost teas and wet soils.
Bhavya reports that the soil sample came from a crop of watermelons that showed signs of stress, grew very little foliage, and produced poorly. The soil had been tilled repeatedly, and several common early-successional plants – which, in an ecological sense, we refer to as weeds – were growing fairly well.
It’s relatively rare to find this microbe in the state in which Bhavya captured it, which of course makes it extra cool. But such a rare sighting also gives us an opportunity to sharpen our soil-detective minds – the more we learn of the intricacies of the microbes we observe in soil, the better we can understand this biological environment as a whole. And therefore we’ll have more pieces of evidence to help us understand a problem and fix it – a problem like the terrible health of those melon plants.
Many problems can be observed on the ground – moisture level, compaction, apparent successional stage – any of which could help explain why the target plant appears to be having such a tough time getting the nutrients it needs. But the lab is also important, and of course any student of the soil foodweb would place a soil sample under the microscope to see which microorganisms are active there – what information can they contribute? When Bhavya did so, he found some interesting microbes that together showed a consistent pattern. There were fungal spores visibly matching those of Fusarium (a common pathogen), along with rotifers, ciliates, and plant-parasitic nematodes – all of which are common to lower-oxygen environments, the opposite of what our productive terrestrial plants need in soil.
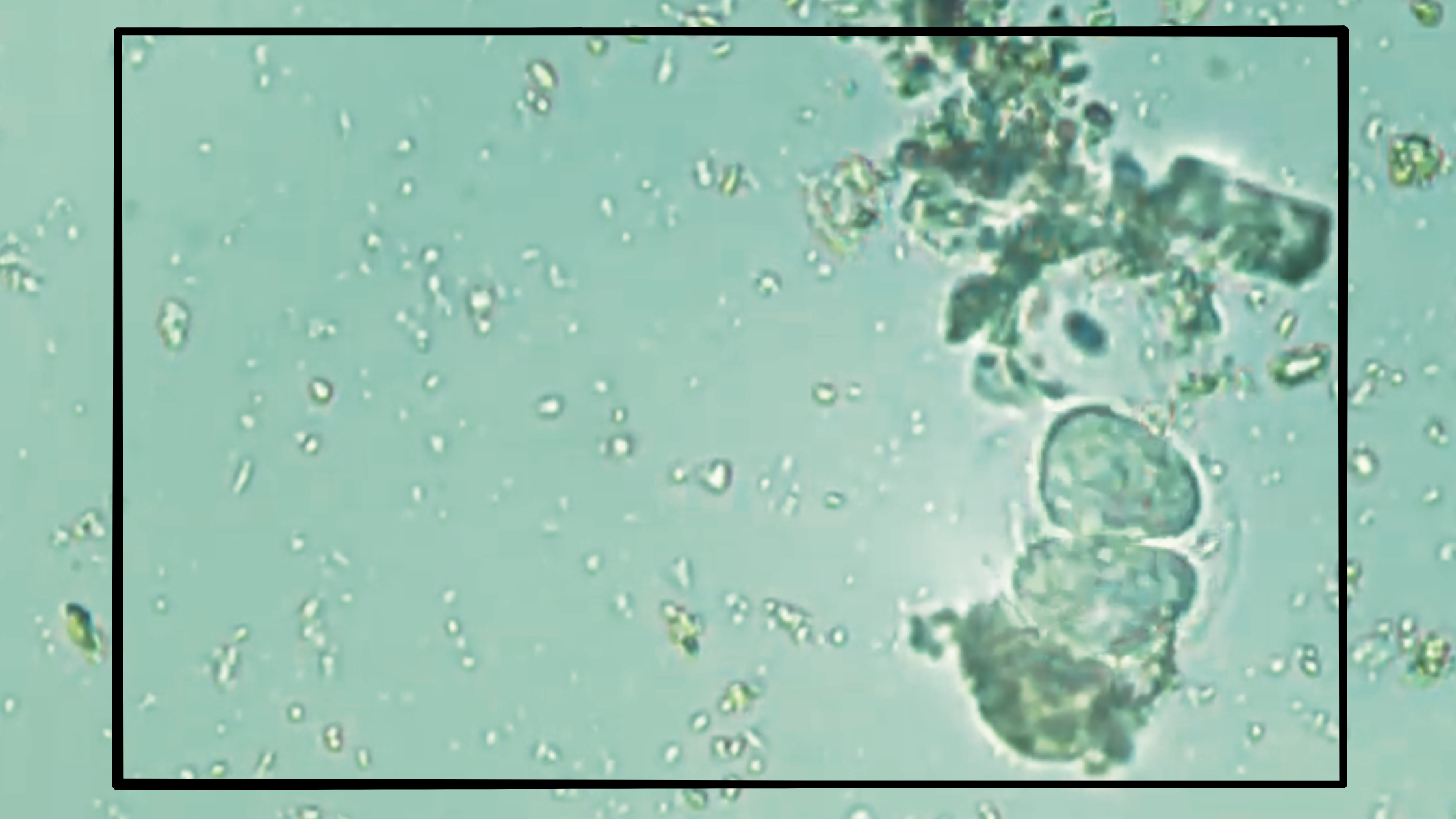
With all this in mind, it’s important to identify the specimen in Bhavya’s video correctly – if it doesn’t fit the pattern, that would be a useful bit of evidence. At first glance, it appears to show amoeba-like qualities. It has a large circular enclosure, sort of like the spherical naked amoeba (without a protective shell) commonly found under elevated oxygen conditions – quite the opposite of what those other specimens, as well as the evidence on the ground, suggest.
However, even if you’ve never seen something like this, you’ll likely realize a couple of things: first, amoebae don’t move nearly that quickly; and second, that idea doesn’t explain the presence of two apparently individual organisms inside the larger membrane.
Given the size (relatively large) and rapid motion, we can conclude with reasonable doubt that the two organisms inside are not amoebae, but ciliates, a broad group of protozoans that dwell in lower-oxygen conditions – exactly the sort of environment that would stress most terrestrial plants.
These ciliates appear to be in the final stage of emergence from a cyst. We tend to think of cysts as only dormancy-related objects (“resting” cysts), created by the organism to wait out unfavorable conditions in a sleep-like state. Once favorable conditions return, many ciliates begin emerging first by forming an excystment vacuole that expands to eventually rupture the cyst. The ciliate escapes surrounded by its endocyst, the membrane that lines the cyst’s inner wall. As the hyperactive ciliate gyrates inside, this membrane soon breaks open, and the ciliate swims off.
But there’s a second reason that ciliates form cysts: for reproduction (the “reproductive” cyst). They need the protection of a hard shell not only for dormancy, but also for undergoing cell division, the process by which single-celled organisms multiply. Here’s a shot showing Colpodids (a family of ciliates that Bhavya’s specimens appear to resemble) having divided not once, but twice, with four ciliates emerging from each endocyst.
So, taken as a clue in the course of SFW work, what are these organisms suggesting? The same thing suggested by a single ciliate emerging from a resting cyst: for them, conditions are good. They would not be active, let alone reproducing, if this weren’t the case. And therefore, conditions may not be so great for the sorts of terrestrial plants that we tend to grow in our gardens and on our farms. Those plants require a soil that isn’t too wet or compacted. How best to restore such conditions in the location where those melons were growing? Every site and project is unique, so it’s best to ask a certified consultant to investigate in person.
A good consultant, as well as a good student, knows to consider all evidence, and to always keep their camera ready. You never know what you’ll find while at the microscope, and how it might contribute not only to your own grasp of the problem at hand, but also to the body of knowledge and understanding we are continually building here at the Soil Food Web Foundation.